All products are handled with strict quality standards to ensure consistent research-grade excellence.
Our checkout is SSL encrypted and completely secure.
Our products are verified by independent third party laboratories to meet quality standards.
All product batches and lots are assigned unique identifiers and tied to publicly posted lab reports.
We are committed to helping our customers. If you have any questions or concerns, please reach out through our Contact Us page.
$34.99 – $54.99Price range: $34.99 through $54.99
| Quantity | Discount | Price |
|---|---|---|
| 5 - 10 | 5% | $52.24 |
| 11 - 20 | 10% | $49.49 |
| 21+ | 15% | $46.74 |
Every batch of our research chemicals and peptides undergoes third-party testing.
*Disclaimer: This product is intended solely for laboratory research purposes. It is not suitable for consumption by humans, nor for medical, veterinary, or household purposes. Kindly review our Terms & Conditions before making a purchase.

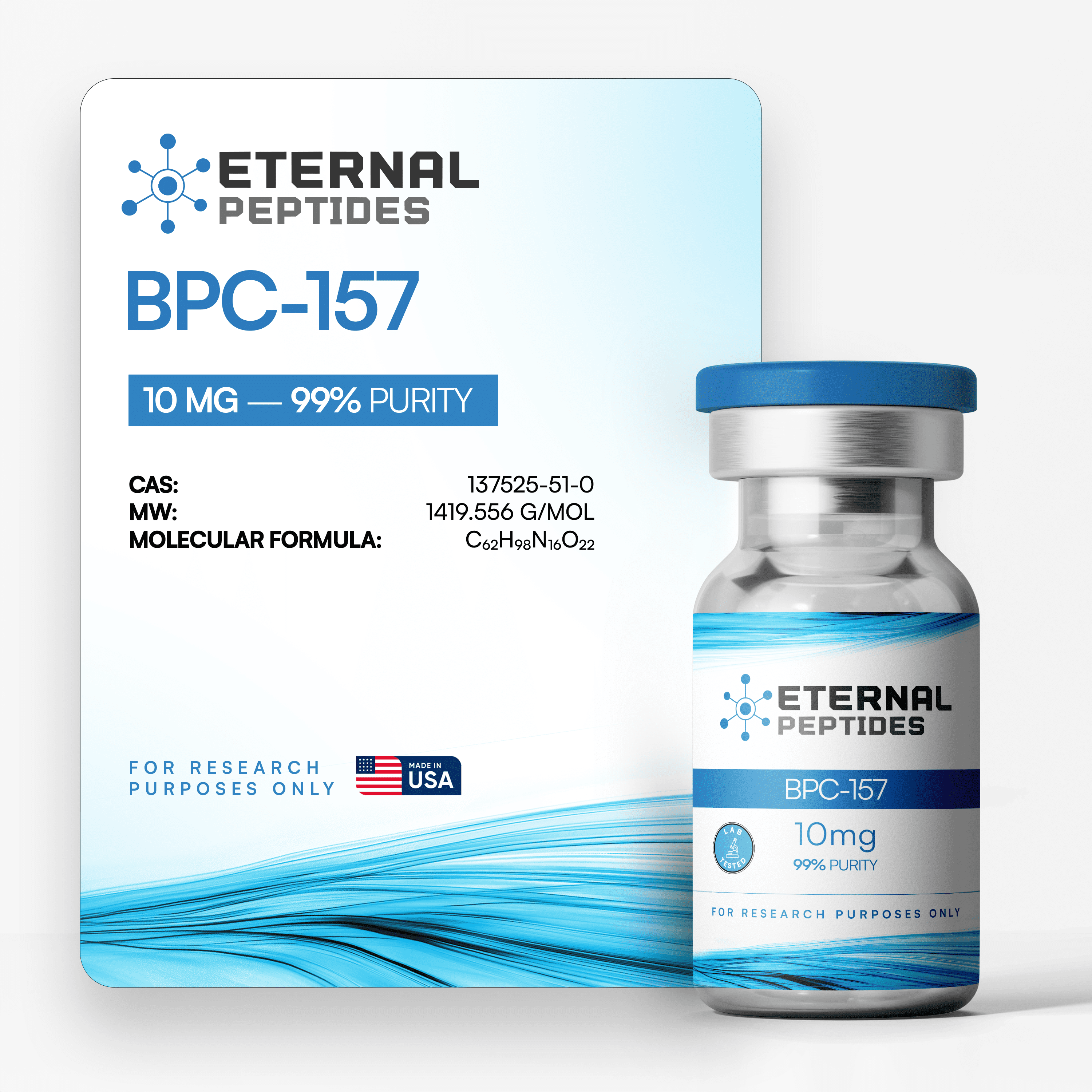
At every step, we prioritize quality by conducting rigorous third-party testing on all our products. These tests focus on five key characteristics- identity, purity, sterility, and endotoxin levels, and heavy metal content-ensuring that each product meets the highest standards of quality with independent third-party Certificates of Analysis (COAS) to verify our commitment to excellence.
*Disclaimer: This product is intended solely for laboratory research purposes. It is not suitable for consumption by humans, nor for medical, veterinary, or household purposes.Kindly review our Terms & Conditions before making a purchase.
Order DSIP 5 mg from Eternal Peptides, a top peptides supplier in the U.S. committed to consistent quality through rigorous third-party testing by leading labs. This high-purity Delta Sleep-Inducing Peptide (DSIP) is used in research exploring sleep regulation, stress response, and neuroprotective mechanisms. Get high-purity DSIP 10mg, verified with Certificates of Analysis and at competitive pricing. Free USPS shipping for orders above $200 with dedicated support. DSIP is sold for research use only.
Delta Sleep-Inducing Peptide (DSIP) is a naturally occurring neuropeptide consisting of nine amino acids (Trp-Ala-Gly-Gly-Asp-Ala-Ser-Gly-Glu). It was first isolated from the cerebral venous blood of rabbits during slow-wave sleep in 1977 by Swiss researchers Schoenenberger and Monnier.
This endogenous peptide is found in various regions of the brain, as well as in peripheral organs and plasma, where it appears to play multifaceted roles in physiological regulation.
DSIP is primarily investigated for its involvement in sleep-wake cycle modulation, stress adaptation, pain perception, and neuroendocrine regulation. Research has explored its potential effects on corticotropin and somatotropin secretion, oxidative stress response, and neuroprotective mechanisms.
Most current findings derive from in vitro studies and animal models, particularly rodent research examining sleep architecture, cortisol levels, and behavioral responses to stress.
DSIP’s small molecular size and relatively simple structure contribute to its stability in solution and ease of handling in laboratory settings, making it well-suited for controlled research applications. Its water solubility also facilitates precise dosing and administration across various experimental protocols.
Delta Sleep-Inducing Peptide (DSIP) operates through multiple neuroendocrine and cellular pathways, though its exact mechanisms remain partially understood. Research suggests DSIP modulates sleep-wake cycles primarily through interactions with GABAergic and serotonergic systems, influencing neurotransmitter release and neuronal excitability in sleep-regulating brain regions[1].
The peptide also appears to affect the hypothalamic-pituitary-adrenal (HPA) axis, demonstrating stress-adaptive properties by modulating corticotropin and cortisol secretion in animal models[2]. Additionally, cellular studies indicate potential antioxidant and cytoprotective effects through regulation of free radical metabolism and lipid peroxidation pathways.
In rodent studies, DSIP administration has been associated with alterations in sleep architecture, particularly increased slow-wave sleep duration and modifications to REM sleep patterns[2]. Animal research has also observed reductions in stress-induced behavioral changes and normalization of stress hormone profiles following chronic stress exposure, alongside potential analgesic effects in pain models.
DSIP’s effects on sleep patterns appear mediated through complex interactions with central nervous system neurotransmitter systems. Animal studies suggest the peptide influences GABAergic transmission, the brain’s primary inhibitory signaling pathway, potentially enhancing chloride channel activity and neuronal hyperpolarization in sleep-promoting regions such as the ventrolateral preoptic nucleus[1].
Research also indicates modulation of serotonergic pathways, with alterations in 5-HT receptor sensitivity and turnover rates observed in rodent brain tissue[3]. Electrophysiological recordings in laboratory animals have documented increased delta wave amplitude and prolonged slow-wave sleep episodes following DSIP administration[4].
These observations have made DSIP a subject of interest in investigations examining circadian rhythm disruption, sleep deprivation models, and age-related sleep disturbances in experimental settings, though translation to human sleep physiology remains incompletely characterized.
DSIP demonstrates notable interactions with the hypothalamic-pituitary-adrenal axis, the body’s central stress response system.
In animal models subjected to various stressors, including restraint stress, cold exposure, and electric shock, DSIP administration has been associated with attenuated corticotropin-releasing hormone (CRH) secretion and reduced plasma corticosterone levels compared to controls[5].
Research suggests this occurs through direct or indirect modulation of hypothalamic CRH neurons and pituitary corticotrophs. Some studies indicate DSIP may enhance negative feedback sensitivity to circulating glucocorticoids, thereby preventing excessive or prolonged stress hormone elevation.
Behavioral assessments in rodents have shown reduced anxiety-like behaviors and improved adaptation to repeated stress following DSIP treatment[6]. These findings have positioned DSIP as a compound of interest in research exploring stress resilience, glucocorticoid dysregulation models, and the neurobiological mechanisms underlying stress-related behavioral changes.
Cellular and tissue-level studies have identified potential antioxidant properties of DSIP, particularly regarding oxidative stress mitigation[7]. In vitro experiments demonstrate that DSIP can reduce lipid peroxidation markers and increase antioxidant enzyme activities, including superoxide dismutase and catalase, in neuronal and hepatic cell cultures exposed to oxidative challenges.
Animal research has shown protective effects against chemically induced oxidative damage in brain tissue, with reduced malondialdehyde formation and preserved mitochondrial function[8]. The mechanism appears to involve modulation of reactive oxygen species (ROS) generation and enhancement of endogenous antioxidant defense systems rather than direct free radical scavenging.
Some studies suggest DSIP may stabilize cellular membranes and reduce calcium influx during oxidative stress, contributing to cytoprotection. These observations have made DSIP relevant to research models investigating ischemia-reperfusion injury, neurotoxicity, and age-related oxidative damage in laboratory settings[8].
It is important to note that the majority of mechanistic data on DSIP derives from in vitro cellular assays and animal studies, primarily in rodents. Controlled human clinical trials investigating DSIP’s mechanisms and physiological effects are extremely limited, and the peptide has not been approved for therapeutic use.
As such, the translation of findings from preclinical models to human physiology remains uncertain, and the specific receptor(s) mediating DSIP’s diverse effects have not been definitively identified. All mechanistic descriptions should be interpreted strictly within a preclinical research context, and findings cannot be extrapolated to human health applications without substantial additional investigation.
DSIP has attracted scientific interest across multiple research domains, including sleep physiology, stress biology, pain mechanisms, and oxidative stress pathways.
The applications discussed below represent observations from preclinical animal models and cellular assays. These findings do not imply any human or veterinary therapeutic benefits, as controlled clinical evidence is lacking.
DSIP is not approved for medical use, and Eternal Peptides does not promote or advocate for any human or veterinary applications.
Animal research has consistently documented alterations in sleep architecture following DSIP administration[9]. Rodent studies using polysomnographic recordings have shown increased slow-wave sleep duration, reduced sleep latency, and modifications to REM sleep distribution.
Electrophysiological assessments reveal enhanced delta wave activity in cortical regions associated with deep sleep. Research in sleep-deprived animal models has demonstrated partial restoration of normal sleep patterns and reduced behavioral deficits associated with sleep restriction.
Some investigations have examined DSIP’s effects in aged rodents, where natural sleep fragmentation occurs, observing improvements in sleep consolidation and reduced nighttime awakenings. These findings have made DSIP valuable for research protocols examining circadian disruption, sleep deprivation consequences, and the neurobiological basis of sleep regulation[10].
In simpler terms, laboratory animals given DSIP tend to fall asleep faster, spend more time in deep sleep phases, and show more stable sleep patterns, particularly useful for studying what happens when sleep is disrupted or reduced.
DSIP has been extensively studied in animal stress models, demonstrating effects on both physiological and behavioral stress responses. Rodents exposed to chronic unpredictable stress and treated with DSIP show attenuated elevations in plasma corticosterone compared to controls, suggesting modulation of HPA axis reactivity[5].
Behavioral assessments reveal reduced anxiety-like behaviors in elevated plus maze and open field tests. Some studies report normalized stress-induced disruptions in neurotransmitter systems, particularly serotonergic and dopaminergic pathways in limbic structures. These applications make DSIP relevant for investigating stress resilience mechanisms, glucocorticoid regulation, and the neurobiological underpinnings of stress-related behavioral changes[11].
Put simply, animals under stressful conditions and given DSIP show lower stress hormone levels and less anxious behavior, helping researchers understand how the body adapts to ongoing stress.
Research has explored DSIP’s effects in various animal pain models with notable observations[12]. In rodent studies using thermal and mechanical nociception tests, DSIP administration has been associated with increased pain thresholds and prolonged latency to pain responses.
Some investigations report synergistic effects when combined with opioid analgesics, potentially reducing required opioid dosages while maintaining analgesic efficacy.
Research in inflammatory pain models shows reduced pain-related behaviors and decreased neuronal activation in pain-processing brain regions. The mechanisms appear distinct from classical opioid pathways, as naloxone does not completely block DSIP’s observed effects. These findings position DSIP as a tool for investigating non-opioid pain modulation pathways and understanding the complex neurobiology of pain perception.
In practical research terms, laboratory animals given DSIP respond less intensely to painful stimuli and show reduced pain-related behaviors, offering insights into alternative pain pathways beyond traditional opioid systems.
Cellular and animal studies have examined DSIP’s potential protective effects against oxidative damage[13]. In vitro experiments using neuronal cultures exposed to oxidative stressors show reduced lipid peroxidation markers and preserved cell viability when treated with DSIP.
These applications make DSIP useful for research investigating oxidative stress mechanisms, cellular defense systems, and potential interventions in oxidative damage models across various tissue types[14].
Simply stated, cells and tissues exposed to oxidative damage show better survival and less harmful byproduct formation when DSIP is present, valuable for studying how organisms defend against cellular stress.
| Property | Details |
| Name | Delta Sleep-Inducing Peptide (DSIP) |
| Sequence | Trp-Ala-Gly-Gly-Asp-Ala-Ser-Gly-Glu (WAGGDASGE) |
| Molecular Weight | 848.8 g/mol |
| Molecular Formula | C₃₅H₄₈N₁₀O₁₅ |
| PubChem CID | 68816 |
| Product Form | Lyophilized powder in sterile 5 mg vials |
| Purity | ≥99% (verified via third-party testing and lot-specific COA) |
| Storage | Store lyophilized powder at -4°F (-20°C); protect from light and moisture |
| Stability | Stable as lyophilized powder for extended periods when stored properly; reconstituted solutions should be used promptly or stored at 2-8°C for short-term use |
| Solubility | Soluble in sterile water or bacteriostatic water; typical reconstitution at 1-2 mg/mL concentration recommended for research applications |
Note: DSIP is supplied as a lyophilized powder requiring reconstitution with an appropriate sterile solvent before use in research protocols. The nonapeptide structure contains both hydrophobic (tryptophan) and charged (aspartic acid, glutamic acid) residues, contributing to its amphipathic properties.
Proper handling and storage of DSIP are essential to maintain peptide integrity, stability, and reproducibility across research protocols. Lyophilized DSIP powder should be stored at -20°C (-4°F) in a freezer, protected from light and moisture to prevent degradation. Keep vials sealed until ready for use, and allow them to reach room temperature before opening to minimize condensation exposure.
Key handling recommendations:
Eternal Peptides provides comprehensive Certificates of Analysis (COAs) for every DSIP production lot, ensuring transparency, traceability, and confidence in product quality. These COAs support reproducible research outcomes and facilitate regulatory compliance for institutional audits and documentation requirements.
Each lot-specific COA includes:
Eternal Peptides partners with independent third-party laboratories, including Janoshik, Finnrick Analytics, and other leading analytical facilities, to conduct rigorous testing that meets or exceeds industry standards. All COAs are accessible through the Lab Tests page on the Eternal Peptides website.
DSIP is supplied strictly for laboratory research purposes only and is not approved by the FDA or any regulatory authority for human use, veterinary use, clinical administration, therapeutic applications, or diagnostic procedures.
The safety, efficacy, and pharmacological properties of DSIP in humans have not been established through controlled clinical trials. This product is intended solely for qualified researchers working in appropriately equipped laboratory facilities under institutional oversight.
Purchasers are solely responsible for ensuring compliance with all applicable federal, state, and local laws, as well as institutional biosafety policies, animal care regulations, and research-use guidelines governing their jurisdiction and research setting.
Eternal Peptides does not advocate, support, or condone any non-research use of this product. By purchasing DSIP, researchers acknowledge their responsibility for lawful, ethical, and compliant use in legitimate scientific investigation.
https://pmc.ncbi.nlm.nih.gov/articles/PMC11498945/
https://pubmed.ncbi.nlm.nih.gov/2839244/
https://pubmed.ncbi.nlm.nih.gov/3620931/
https://www.sciencedirect.com/topics/medicine-and-dentistry/delta-sleep-inducing-peptide
https://pubmed.ncbi.nlm.nih.gov/2995861/
https://www.mdpi.com/1420-3049/26/17/5173
https://pubmed.ncbi.nlm.nih.gov/25898718/
https://pubmed.ncbi.nlm.nih.gov/12668217/
https://www.sciencedirect.com/science/article/abs/pii/S0024320518304727
https://pubmed.ncbi.nlm.nih.gov/3792404/
https://pubmed.ncbi.nlm.nih.gov/8597403/
https://pubmed.ncbi.nlm.nih.gov/2853064/
https://link.springer.com/article/10.1134/S2079057011040035
https://pubmed.ncbi.nlm.nih.gov/11421812/
About the Author:
Dr. Sony Sherpa, MBBS, MD
is a board-certified clinician with a background in emergency medicine and clinical practice. She specializes in medical research analysis, ensuring that product information is grounded in evidence-based medicine and strictly adheres to the latest scientific standards in peptide research and recovery.