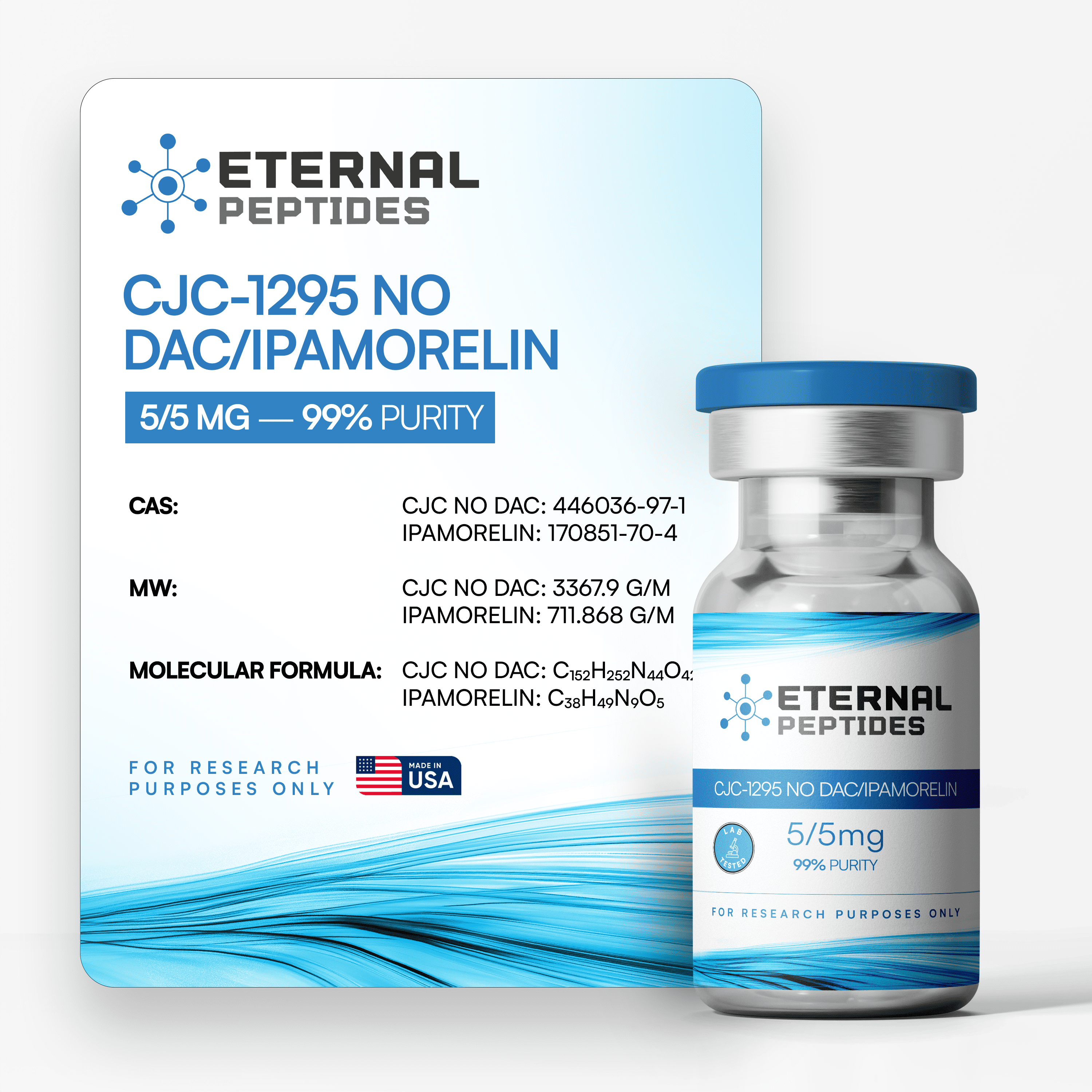
All products are handled with strict quality standards to ensure consistent research-grade excellence.
Our checkout is SSL encrypted and completely secure.
Our products are verified by independent third party laboratories to meet quality standards.
All product batches and lots are assigned unique identifiers and tied to publicly posted lab reports.
We are committed to helping our customers. If you have any questions or concerns, please reach out through our Contact Us page.
$39.99 – $69.99Price range: $39.99 through $69.99
| Quantity | Discount | Price |
|---|---|---|
| 5 - 10 | 5% | $66.49 |
| 11 - 20 | 10% | $62.99 |
| 21+ | 15% | $59.49 |
Every batch of our research chemicals and peptides undergoes third-party testing.
*Disclaimer: This product is intended solely for laboratory research purposes. It is not suitable for consumption by humans, nor for medical, veterinary, or household purposes. Kindly review our Terms & Conditions before making a purchase.

Always quality-tested, verified with third party COA’s
At every step, we prioritize quality by conducting rigorous third-party testing on all our products. These tests focus on five key characteristics- identity, purity, sterility, and endotoxin levels, and heavy metal content-ensuring that each product meets the highest standards of quality with independent third-party Certificates of Analysis (COAS) to verify our commitment to excellence.
*Disclaimer: This product is intended solely for laboratory research purposes. It is not suitable for consumption by humans, nor for medical, veterinary, or household purposes.Kindly review our Terms & Conditions before making a purchase.
GLP-1 (S) 10mg is a synthetic glucagon-like peptide-1 analog supplied for research into GLP-1 receptor signaling, metabolic pathway modeling, and receptor-binding dynamics. Eternal Peptides provides GLP-1 (S) in lyophilized form to preserve stability and activity during storage and shipping. Each lot is independently tested for purity and identity by Janoshik Analytical, with a Certificate of Analysis available for review. Get GLP-1 (S) peptide shipped securely via USPS, free for orders over $200. Sold strictly for research use only.
GLP-1 (S) is a synthetic analog of glucagon-like peptide-1 (GLP-1), an incretin hormone encoded within the proglucagon gene and produced primarily by enteroendocrine L-cells in the distal small intestine and colon[1]. Like GLP-2, GLP-1 is a post-translational processing product of proglucagon, with the specific cleavage pattern in intestinal L-cells yielding both GLP-1 and GLP-2 as co-secreted products in response to nutrient ingestion, particularly fat and carbohydrate.
A second site of GLP-1 production exists in proglucagon-expressing neurons of the brainstem, where it is thought to contribute to central regulation of food intake and energy balance independently of its peripheral metabolic roles.
Native GLP-1 exists in two biologically active truncated forms, GLP-1(7-36) amide and GLP-1(7-37), both of which bind the GLP-1 receptor (GLP-1R), a class B G protein-coupled receptor expressed across multiple tissues including pancreatic beta cells, the gastrointestinal tract, cardiac muscle, kidney, and regions of the central nervous system.
In pancreatic beta cells, GLP-1R activation couples to Gs-mediated cAMP elevation, which potentiates glucose-stimulated insulin secretion, a glucose-dependent mechanism that has made GLP-1R signaling a subject of sustained interest in metabolic research[2].
Receptor activation also suppresses glucagon release from alpha cells, slows gastric emptying, and engages central satiety pathways, collectively producing a coordinated metabolic response to nutrient exposure.
A significant practical limitation of native GLP-1 in experimental settings is its extremely short half-life in circulation, on the order of one to two minutes, due to rapid N-terminal cleavage by dipeptidyl peptidase-4 (DPP-4), the same enzyme that inactivates GLP-2[4]. This degradation renders native GLP-1 poorly suited to experimental paradigms requiring sustained receptor engagement.
Synthetic analogs such as GLP-1 (S) are designed to address this by incorporating structural modifications that confer resistance to DPP-4 cleavage, extending functional half-life and enabling more controlled study of receptor activation kinetics, downstream signaling dynamics, and dose-response relationships in laboratory systems.
GLP-1 (S) is studied as an agonist of the GLP-1 receptor (GLP-1R), meaning its primary mechanism is receptor binding and activation rather than any direct nutritional or substrate effect. What makes GLP-1R signaling particularly useful as a research tool is that its effects are broad and coordinated, spanning pancreatic function, gastrointestinal motility, and central appetite regulation, allowing investigators to use a single receptor-level intervention to probe multiple interconnected aspects of metabolic biology.
The same receptor biology underpins widely studied clinical programs examining GLP-1R agonists in cardiometabolic contexts, including landmark trials such as LEADER and SUSTAIN-6, which have helped frame the questions that preclinical GLP-1 research continues to investigate mechanistically.
GLP-1R is a class B G protein-coupled receptor, meaning it primarily signals through the Gs pathway upon activation. When GLP-1 (S) binds GLP-1R, the receptor triggers an increase in intracellular cyclic AMP (cAMP), which activates two principal downstream effectors: protein kinase A (PKA) and exchange protein directly activated by cAMP (Epac)[4].
In pancreatic beta cells, where GLP-1R expression is well established, this cAMP elevation amplifies the insulin secretory response to glucose, but critically, only when glucose is already elevated. This glucose-dependence is a defining feature of GLP-1R-mediated insulin secretion and distinguishes it mechanistically from glucose-independent secretagogues. PKA and Epac act through partially distinct routes to enhance this response: PKA phosphorylates ion channel proteins and secretory machinery components that facilitate insulin granule exocytosis, while Epac influences intracellular calcium handling and vesicle priming.
Together, these signals increase the sensitivity and magnitude of insulin release in proportion to the prevailing glucose concentration.
Beyond insulin secretion, GLP-1R activation in alpha cells suppresses glucagon release, an effect that complements the insulin response by reducing hepatic glucose output, and has been associated in cell-based models with signaling linked to beta cell survival and, in some rodent studies, beta cell proliferation, though the latter finding has been more difficult to demonstrate in human islet preparations[5].
GLP-1R signaling also exhibits what researchers describe as signaling bias, which is the observation that different ligands binding the same receptor can preferentially engage different downstream pathways, such as cAMP/PKA versus beta-arrestin-mediated internalization. This has made GLP-1R a useful model system for studying biased agonism as a broader pharmacological concept, with GLP-1 analogs serving as reference compounds against which signaling profiles can be compared.
GLP-1R is expressed not only in the pancreas but in vagal afferent neurons, the brainstem, and the hypothalamus, and activation of these central and peripheral neural populations produces effects on appetite and gastrointestinal function that are distinct from the pancreatic insulin secretion story[6].
In animal models, GLP-1R activation slows gastric emptying, a mechanical effect that blunts the speed of postprandial glucose absorption and contributes to the overall glucose-lowering response. This is studied in laboratory settings using gastric motility assays and gastrointestinal transit measurements.
At the level of the central nervous system, GLP-1R-expressing neurons in the brainstem nucleus tractus solitarius (NTS) and hypothalamic regions including the arcuate nucleus receive both direct peptide signaling and vagally relayed input from the gut. Activation of these circuits in rodent models reduces food intake and meal size, and has been associated with changes in the expression of hypothalamic neuropeptides involved in energy balance regulation, including neuropeptide Y (NPY) and pro-opiomelanocortin (POMC).
These neuroendocrine readouts are commonly used endpoints in preclinical studies examining feeding behavior and meal patterning in response to GLP-1R agonism.
The mechanistic picture described here draws on a substantial body of in vitro and animal model research accumulated over several decades of GLP-1 biology. However, controlled human evidence for GLP-1 (S) specifically as a research compound is limited, and the complexity of GLP-1R’s multi-tissue signaling profile means that findings from isolated cell systems or rodent models do not always translate directly to human physiology.
The glucose-dependent nature of GLP-1R-mediated insulin secretion, the species differences in islet architecture and receptor expression, and the distinct central nervous system circuitry involved in human appetite regulation are all factors that warrant caution when extrapolating preclinical data. These findings should be interpreted primarily within the in vitro and animal model context in which they were obtained.
Preclinical investigations of GLP-1 receptor agonists, including synthetic GLP-1 analogs such as GLP-1 (S), primarily examine metabolic regulation, energy balance, pancreatic islet biology, cardiovascular signaling, and neuroprotection.
These observations are derived largely from in vitro systems and animal models. They do not imply human or veterinary benefit, and this compound is not approved for therapeutic use. Eternal Peptides supplies GLP-1 (S) strictly for laboratory research purposes.
In rodent and cellular studies, GLP-1 receptor activation enhances glucose-dependent insulin secretion and suppresses glucagon release from pancreatic alpha cells, together producing a coordinated reduction in blood glucose that has made this receptor system one of the most studied targets in metabolic biology[2].
Researchers use GLP-1 analogs to investigate beta-cell stimulus-secretion coupling, intracellular cAMP dynamics, and glucose tolerance under controlled experimental conditions, often employing isolated islet preparations or GLP-1R-expressing cell lines to dissect the specific contributions of PKA and Epac signaling to the secretory response. These models help clarify how incretin signaling integrates with other inputs to regulate pancreatic responsiveness and systemic glucose handling across varying metabolic states.
In short, scientists use GLP-1 (S) to observe how pancreatic cells respond to glucose when GLP-1 receptor pathways are active, and to map the intracellular signals that connect receptor activation to insulin release.
GLP-1 signaling is widely studied in animal models of appetite regulation and energy expenditure, reflecting the receptor’s expression across both peripheral tissues and central nervous system circuits involved in feeding behavior[3].
Central and peripheral GLP-1 receptor activation has been associated with reduced food intake, delayed gastric emptying, and altered hypothalamic neuropeptide signaling, including changes in NPY and POMC expression, that collectively shift the balance toward reduced caloric intake and increased satiety signaling.
These effects are typically evaluated in rodent feeding studies and metabolic cage experiments measuring caloric intake, meal patterning, gastric transit, and body mass trends over defined experimental periods, providing a controlled framework for understanding how GLP-1R agonism interacts with the broader neural and hormonal architecture of energy balance.
Preclinical research also explores GLP-1 receptor activity in cardiovascular tissues, motivated in part by GLP-1R expression in cardiac muscle, vascular endothelium, and smooth muscle cells across multiple species[7]. Experimental models suggest that GLP-1 signaling may influence endothelial nitric oxide production, inflammatory marker expression, and myocardial substrate metabolism, effects that have been investigated using isolated vessel preparations, cardiomyocyte cell cultures, and animal models of metabolic stress or ischemia-reperfusion injury.
While these findings have generated interest in the cardiovascular dimensions of GLP-1R biology, the specific signaling mechanisms involved and their relevance beyond the experimental systems studied remain areas of active preclinical investigation.
GLP-1 receptors are expressed in multiple brain regions, including the hypothalamus, brainstem, hippocampus, and cortex, and animal studies investigate their role in neuronal survival pathways, synaptic plasticity, and neuroinflammatory modulation. Laboratory models of neurodegeneration and metabolic stress often incorporate GLP-1 analogs to assess activation of intracellular signaling cascades such as PI3K/Akt and MAPK/ERK pathways, which are associated with cellular resilience and the regulation of inflammatory mediators including NF-κB[8].
Some rodent studies have also examined GLP-1R’s potential interactions with dopaminergic and reward-related circuits, reflecting broader interest in how metabolic peptide signaling intersects with neurological function, though this work remains at an early preclinical stage.
In other words, researchers use GLP-1 (S) to study how brain cells respond to metabolic and inflammatory stress in experimental models, with a focus on the signaling patterns that govern cell survival and inflammation rather than any treatment-related outcome.
| Property | Value |
| Name | GLP-1 (S) synthetic glucagon-like peptide-1 analogue |
| Sequence | Modified peptide based on human GLP-1 (7-37) backbone with non-native substitutions to enhance stability (actual proprietary sequence varies) |
| Molecular Weight | 4113.641 g/mol |
| Molecular Formula | C187H291N45O59 |
| PubChem CID | 56843331 |
| Product Form | Lyophilized powder for reconstitution |
| Purity | ≥99% as verified by lot-specific Certificates of Analysis |
| Solubility | Soluble in bacteriostatic water or appropriate buffered aqueous solutions after reconstitution; avoid organic solvents until dissolved |
| Stability | Lyophilized format supports enhanced shelf life when stored refrigerated or frozen; reconstituted solutions should be used promptly |
| Handling Notes | Handle under inert conditions for prolonged storage of reconstituted material; protect from repeated freeze–thaw cycles |
Note: This GLP-1 (S) product is offered for research use only and not intended for human or veterinary applications. Sequence and modifications are designed to improve receptor engagement and metabolic resistance compared to native GLP-1, with structural data aligned to related analogues in public chemical databases.
Proper handling and storage are essential to preserve structural integrity and experimental consistency when working with GLP-1 (S). As a lyophilized synthetic peptide analogue, it should be protected from heat, moisture, and repeated temperature fluctuations prior to and after reconstitution.
Each lot of GLP-1 (S) is supported by a lot-specific Certificate of Analysis (COA) to ensure transparency, traceability, and reproducibility in laboratory research. Eternal Peptides partners with independent third-party analytical laboratories, including Janoshik and Finnrick Analytics, to verify identity and quality prior to release.
COAs for GLP-1 (S) typically include:
All COAs are lot-specific, fully traceable, and accessible through the Lab Tests page. This documentation supports internal validation, audit readiness, and consistent batch-to-batch performance in research environments.
GLP-1 (S) is supplied strictly for laboratory research use only. It is not approved for human or veterinary consumption, clinical administration, therapeutic application, or diagnostic procedures of any kind. Safety and efficacy in humans have not been established.
This material must be handled exclusively by qualified professionals in controlled research settings. Purchasers are responsible for ensuring compliance with all applicable local, state, and federal regulations, as well as institutional biosafety policies governing research chemicals.
Any misrepresentation of intended use, including purchase for prohibited applications, may result in regulatory or legal consequences.
About the Author:
Dr. Sony Sherpa, MBBS, MD
is a board-certified clinician with a background in emergency medicine and clinical practice. She specializes in medical research analysis, ensuring that product information is grounded in evidence-based medicine and strictly adheres to the latest scientific standards in peptide research and recovery.