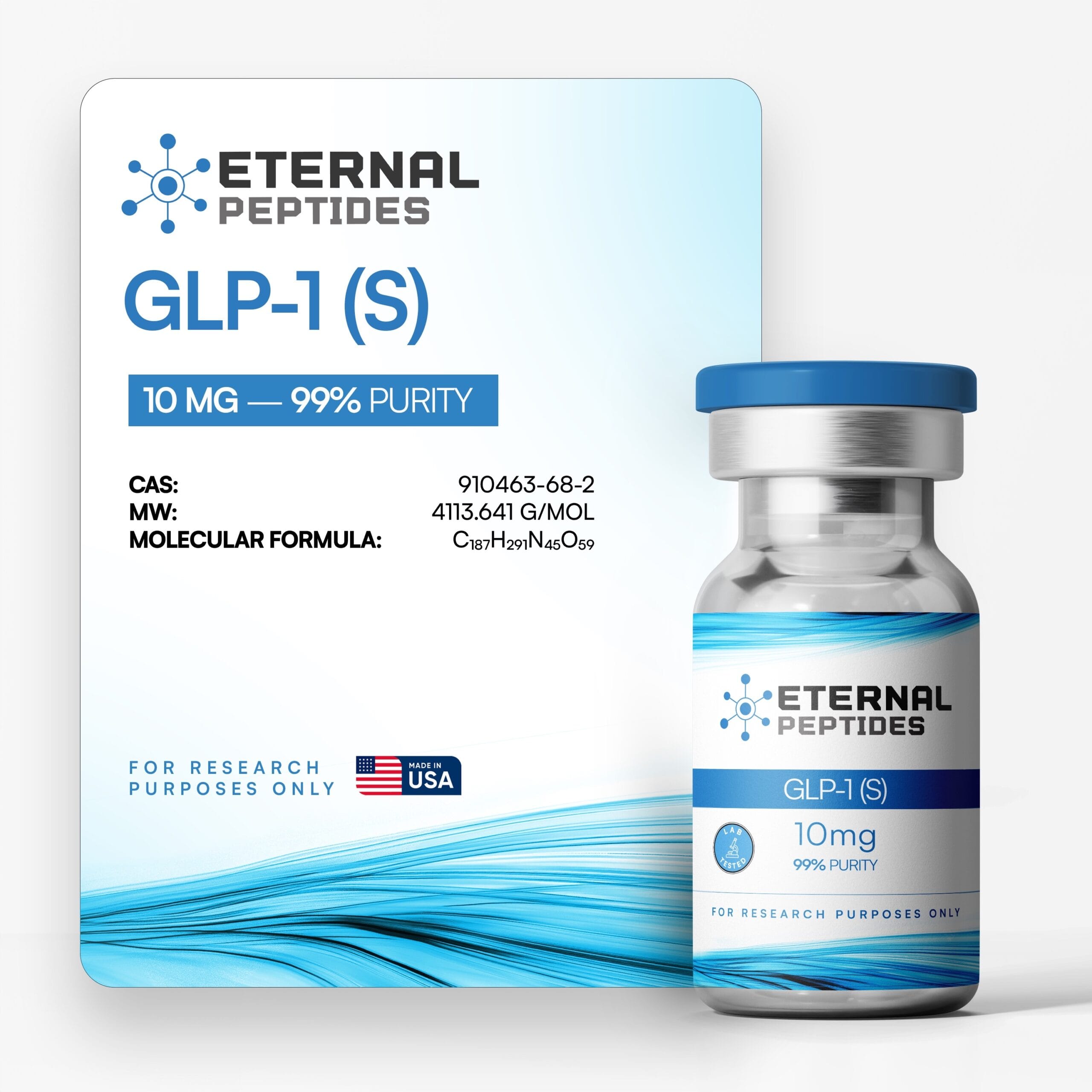
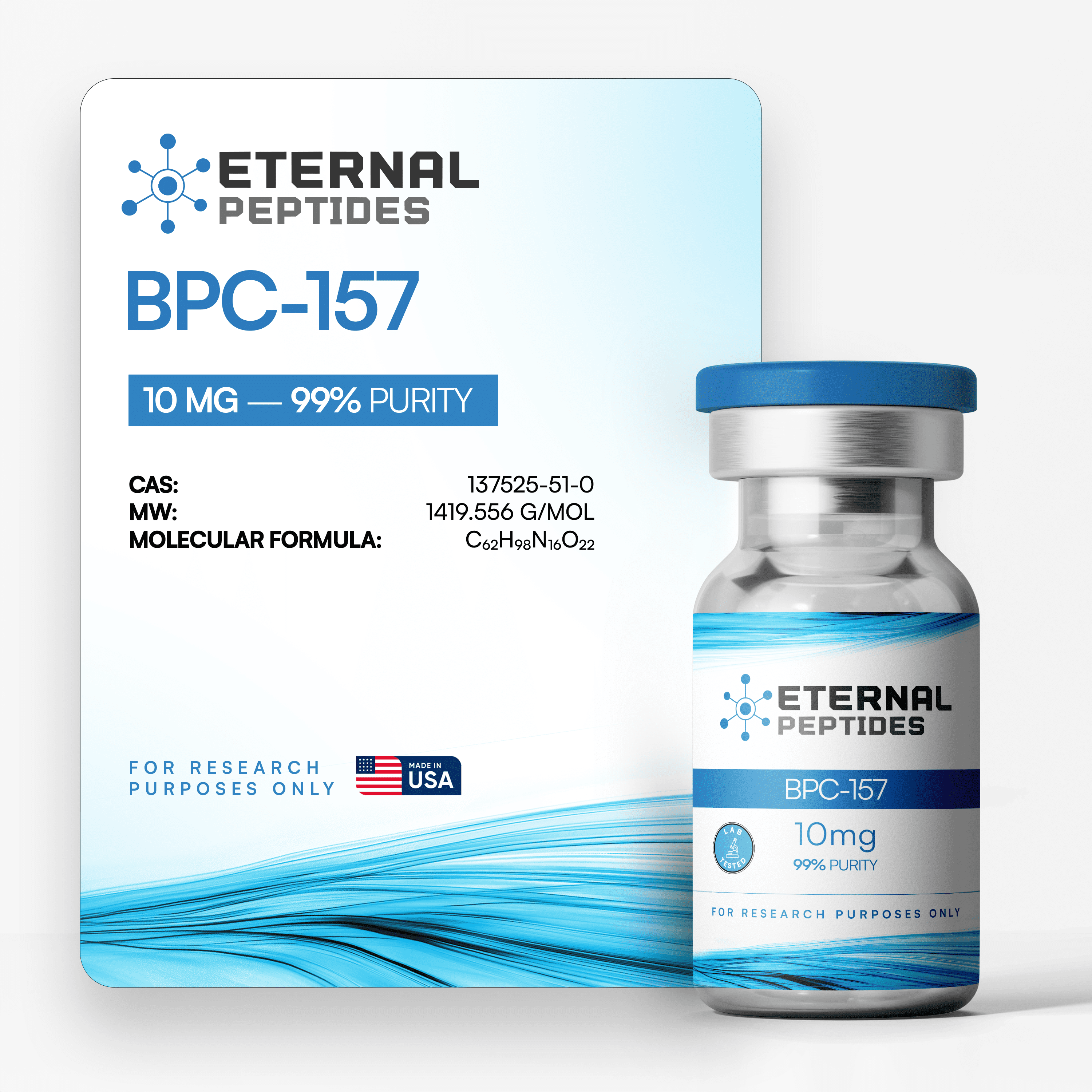
All products are handled with strict quality standards to ensure consistent research-grade excellence.
Our checkout is SSL encrypted and completely secure.
Our products are verified by independent third party laboratories to meet quality standards.
All product batches and lots are assigned unique identifiers and tied to publicly posted lab reports.
$100.00 Original price was: $100.00.$89.99Current price is: $89.99.
| Quantity | Discount | Price |
|---|---|---|
| 5 - 10 | 5% | $85.49 |
| 11 - 20 | 10% | $80.99 |
| 21+ | 15% | $76.49 |
Every batch of our research chemicals and peptides undergoes third-party testing.
*Disclaimer: This product is intended solely for laboratory research purposes. It is not suitable for consumption by humans, nor for medical, veterinary, or household purposes. Kindly review our Terms & Conditions before making a purchase.

Always quality-tested, verified with third party COA’s
At every step, we prioritize quality by conducting rigorous third-party testing on all our products. These tests focus on five key characteristics- identity, purity, sterility, and endotoxin levels, and heavy metal content-ensuring that each product meets the highest standards of quality with independent third-party Certificates of Analysis (COAS) to verify our commitment to excellence.
*Disclaimer: This product is intended solely for laboratory research purposes. It is not suitable for consumption by humans, nor for medical, veterinary, or household purposes.Kindly review our Terms & Conditions before making a purchase.
Shop Glutathione 1500mg from Eternal Peptides, where rigorous third-party testing by independent analytical labs ensures exceptional batch-to-batch consistency for reliable research outcomes. Glutathione is a tripeptide antioxidant designed for research in oxidative stress, cellular detoxification, and immune function studies in its role as the body’s master antioxidant. Buy high-purity verified Glutathione1500mg with free USPS priority shipping on orders over $200. Sold for research use only.
Glutathione (GSH) is an endogenous tripeptide composed of three amino acids: glutamate, cysteine, and glycine (γ-L-Glutamyl-L-cysteinyl-glycine). It was first isolated from yeast in 1888 by French biochemist J. de Rey-Pailhade, who initially termed it philothion (Greek for “sulfur-loving”).
Glutathione was later structurally characterized in the 1920s by Sir Frederick Gowland Hopkins, who elucidated its critical role in cellular redox homeostasis and was awarded the Nobel Prize in part for this work.
As the most abundant non-protein thiol in mammalian cells, glutathione exists naturally in virtually all living organisms and is synthesized intracellularly through ATP-dependent enzymatic reactions involving glutamate-cysteine ligase and glutathione synthetase.
Available scientific literature extensively investigates glutathione for its master antioxidant properties, detoxification mechanisms, immune modulation, mitochondrial function, and protection against oxidative stress. Research spans cellular redox signaling, xenobiotic metabolism, aging processes, neurodegenerative disease models, and hepatic function, with substantial findings from in vitro cellular assays, animal models, and human clinical studies examining oxidative stress conditions.
Synthetic glutathione offers far greater consistency and experimental control than naturally produced glutathione. In vivo glutathione levels fluctuate with diet, stress, disease state, and circadian rhythms, which introduces variability into studies. Synthetic GSH, by contrast, provides a fixed, defined tripeptide composition (γ-glutamyl-cysteinyl-glycine) with known purity and concentration, making it much easier to standardize dosing, reproduce results, and isolate glutathione-specific effects without confounding biological regulation.
Eternal Peptide’s synthetic glutathione offers high purity (≥98%), excellent aqueous solubility, and consistent batch quality essential for reproducible research outcomes.
Glutathione functions as the cell’s primary antioxidant defense system through direct reactive oxygen species (ROS) neutralization[1]. It serves as a cofactor for glutathione peroxidase and glutathione S-transferase enzymes, and maintains cellular redox balance via thiol-disulfide exchange reactions.
The tripeptide participates in Phase II detoxification pathways by conjugating with electrophilic compounds, modulates immune cell function through effects on T-cell proliferation and cytokine production, and protects mitochondrial integrity by scavenging free radicals at their primary generation sites.
Cellular studies demonstrate that glutathione depletion increases susceptibility to oxidative damage, while supplementation in animal models shows protective effects against toxin-induced injury, enhanced immune responses, and improved mitochondrial function[2].
Similarly, research in hepatocyte cultures reveals glutathione’s essential role in xenobiotic metabolism, while neuronal cell assays demonstrate neuroprotective effects under oxidative stress conditions[3].
Glutathione neutralizes reactive oxygen species through direct chemical reduction, with the cysteine residue’s sulfhydryl group (-SH) serving as the reactive site[1]. This thiol group donates electrons to hydrogen peroxide, lipid peroxides, and other oxidants, converting them to less harmful molecules while glutathione itself oxidizes to form glutathione disulfide (GSSG).
The enzyme glutathione reductase then regenerates reduced glutathione using NADPH, maintaining the GSH/GSSG ratio critical for cellular health (typically 100:1 in healthy cells).
Studies in oxidatively stressed cell cultures demonstrate that maintaining adequate glutathione levels prevents lipid peroxidation, protein oxidation, and DNA damage. Animal models exposed to oxidative stressors like paraquat or carbon tetrachloride show significantly reduced tissue damage when glutathione availability is maintained, with corresponding decreases in markers of oxidative injury such as malondialdehyde and 8-hydroxy-2′-deoxyguanosine[4].
Glutathione serves as an essential cofactor for the selenoenzyme glutathione peroxidase (GPx), which catalyzes the reduction of hydrogen peroxide and organic hydroperoxides to water and corresponding alcohols[5].
This enzymatic system provides highly efficient peroxide detoxification, with GPx demonstrating substrate specificity for lipid peroxides that protects membrane integrity. Additionally, glutathione functions as a substrate for glutathione S-transferases (GSTs), a superfamily of enzymes that conjugate glutathione to electrophilic compounds during Phase II detoxification.
Research using hepatocyte cultures demonstrates that GST-mediated glutathione conjugation represents a critical pathway for metabolizing environmental toxins, pharmaceutical compounds, and endogenous metabolic byproducts[6].
Animal studies show that depleting cellular glutathione through buthionine sulfoximine administration dramatically increases susceptibility to acetaminophen hepatotoxicity, while maintaining adequate levels provides dose-dependent protection against liver injury[7].
Glutathione plays a central role in Phase II hepatic detoxification by forming conjugates with electrophilic metabolites generated during Phase I cytochrome P450 reactions[6]. These glutathione conjugates are more water-soluble and can be further processed into mercapturic acids for renal excretion, representing a major elimination pathway for xenobiotics.
In vitro studies using liver microsomes demonstrate that glutathione availability directly influences the detoxification capacity for compounds ranging from environmental pollutants like benzene metabolites to pharmaceutical agents[8].
The tripeptide also detoxifies heavy metals through direct chelation, with the thiol groups binding mercury, lead, and cadmium to facilitate their removal[9]. Animal research using toxicant exposure models consistently shows that glutathione depletion increases organ toxicity, while pre-treatment or co-administration with glutathione precursors reduces tissue damage and mortality across diverse toxicological challenges.
Glutathione significantly influences immune cell function, particularly T-lymphocyte proliferation, differentiation, and cytokine production. The intracellular GSH/GSSG ratio regulates T-cell receptor signaling, with reduced glutathione promoting Th1 responses and oxidized forms favoring Th2 polarization[10].
Studies in isolated lymphocyte cultures reveal that glutathione depletion impairs T-cell proliferation following antigenic stimulation, while restoration normalizes immune responses. Natural killer cell cytotoxicity also depends on adequate glutathione levels, with in vitro assays demonstrating reduced killing capacity under glutathione-depleted conditions[11][12].
Animal models of immune challenge show that maintaining glutathione status supports more robust antibody responses, enhanced pathogen clearance, and improved survival in infection models. Additionally, glutathione modulates dendritic cell maturation and antigen presentation capacity, influencing both innate and adaptive immune coordination in experimental systems.
Mitochondria contain a distinct glutathione pool separate from cytosolic reserves, with this mitochondrial glutathione being critical for protecting respiratory chain complexes from oxidative damage.
Research using isolated mitochondria demonstrates that glutathione depletion increases electron leak from the respiratory chain, accelerating ROS generation and triggering mitochondrial permeability transition, which is a key step in apoptotic cell death[13].
Cellular studies show that glutathione protects mitochondrial DNA from oxidative modifications and maintains membrane potential stability during metabolic stress[14]. In aged animal models, mitochondrial glutathione levels decline significantly, correlating with reduced respiratory capacity, increased oxidative damage, and impaired ATP production.
Conversely, interventions that maintain mitochondrial glutathione in experimental models preserve respiratory function, reduce age-related mitochondrial dysfunction, and protect against toxin-induced energy failure in tissues with high metabolic demands like cardiac and neuronal cells[15].
Glutathione’s biochemical mechanisms are extensively characterized in cellular and animal research, and some human clinical studies have examined glutathione supplementation in specific contexts. However, the bioavailability and systemic effects of exogenous glutathione administration in humans remain subjects of ongoing investigation.
Most mechanistic insights derive from in vitro enzymatic assays, cell culture systems, and rodent models where glutathione metabolism and tissue distribution can be precisely controlled and measured. Direct translation of dosing strategies, pharmacokinetics, and efficacy from these preclinical systems to human applications requires careful consideration of species differences in glutathione synthesis rates, tissue uptake mechanisms, and redox regulatory pathways.
Researchers should interpret findings within appropriate preclinical contexts and recognize that observed protective effects in controlled experimental models do not automatically establish clinical utility or safety profiles for human populations.
Glutathione research encompasses diverse applications, but we emphasize that these findings represent preclinical and experimental research outcomes and do not imply any established human therapeutic benefits or veterinary applications.
Glutathione is not approved by regulatory agencies such as the FDA for disease treatment or prevention, and Eternal Peptides does not promote, advocate for, or support any human consumption or clinical applications of this compound.
Animal studies consistently demonstrate that glutathione administration provides significant protection against hepatotoxic agents including acetaminophen, carbon tetrachloride, alcohol, and various pharmaceutical compounds[16].
Research using rodent models of drug-induced liver injury shows that maintaining hepatic glutathione levels reduces transaminase elevation, decreases histological damage scores, and improves survival rates compared to depleted controls.
Mechanistic studies reveal that glutathione enhances Phase II conjugation reactions, facilitates toxin elimination, and prevents lipid peroxidation in hepatocyte membranes[17]. Isolated hepatocyte cultures exposed to toxic metabolites demonstrate dose-dependent cytoprotection when glutathione availability is maintained, with corresponding reductions in lactate dehydrogenase release and caspase activation markers.
In short, lab animals given liver-damaging substances show less tissue injury and better liver function when glutathione levels are maintained, with liver cells in culture dishes surviving toxic exposures more effectively in glutathione’s presence.
Cellular assays across diverse tissue types demonstrate that glutathione depletion dramatically increases susceptibility to oxidative damage from hydrogen peroxide, superoxide, and hydroxyl radicals, while supplementation provides concentration-dependent protection.
Research using oxidatively stressed cell lines shows preserved mitochondrial membrane potential, reduced DNA strand breaks, decreased protein carbonylation, and maintained cellular viability in glutathione-enriched conditions[18].
Animal models subjected to ischemia-reperfusion injury in organs such as the heart, brain, kidney, and liver exhibit reduced infarct sizes, improved functional recovery, and decreased inflammatory marker expression when glutathione status is optimized prior to or immediately following the ischemic event[1].
These protective effects correlate with reduced lipid peroxidation products, preserved antioxidant enzyme activities, and decreased apoptotic cell death in affected tissues.
In other words, cells and tissues exposed to damaging oxygen radicals survive better and maintain normal function when glutathione is present, with organs recovering more completely from blood flow interruptions in laboratory models where glutathione levels are maintained.
Longitudinal studies in various model organisms including yeast, nematodes, and rodents reveal strong correlations between glutathione levels and lifespan, with age-related glutathione decline associated with increased oxidative damage, mitochondrial dysfunction, and cellular senescence[19].
Research using aged animal models demonstrates that interventions maintaining glutathione status preserve cognitive function, reduce age-related pathology markers, and extend healthspan metrics including physical performance and metabolic flexibility[20].
Caloric restriction paradigms, which are known to extend lifespan across species, consistently elevate tissue glutathione levels, and some research suggests glutathione maintenance may mediate portions of caloric restriction’s beneficial effects. Also, cellular senescence studies show that maintaining glutathione delays replicative senescence onset and reduces senescence-associated secretory phenotype markers in cultured cells approaching their proliferative limits.
More simply, research shows that as laboratory animals age, their glutathione levels naturally decline alongside deteriorating health markers. Interventions that maintain glutathione correlate with better preservation of youthful function and extended lifespan in experimental models.
Neurodegenerative disease models including Parkinson’s, Alzheimer’s, and ALS demonstrate characteristic glutathione depletion in affected brain regions, with this depletion preceding overt neuronal loss in many experimental paradigms.
Research using neurotoxin-induced Parkinson’s models shows that maintaining brain glutathione levels provides significant protection against dopaminergic neuron death, preserves motor function, and reduces α-synuclein aggregation[21].
Similarly, Alzheimer’s disease models reveal that glutathione status influences amyloid-beta toxicity and tau phosphorylation, with glutathione-depleted neurons showing increased vulnerability to these pathological proteins[22]. Neuronal culture studies demonstrate that glutathione protects against excitotoxicity, reduces mitochondrial dysfunction, and maintains synaptic integrity under various stress conditions relevant to neurodegenerative processes.
In summary, brain tissue from animal models of neurological diseases shows low glutathione alongside nerve cell damage, while maintaining glutathione levels helps protect brain cells from toxins and disease-related proteins in laboratory studies.
| Property | Details |
| Name | Glutathione L-Glutathione (Reduced form, GSH) |
| Sequence | γ-L-Glutamyl-L-cysteinyl-glycine (Glu-Cys-Gly) |
| Molecular Formula | C10H17N3O6S |
| Molecular Weight | 307.33 g/mol |
| PubChem CID | 124886 |
| Product Form | Lyophilized powder in 1500 mg vials |
| Purity | ≥99%, verified via third-party laboratory analysis with lot-specific Certificates of Analysis (COA) available |
| Solubility | Highly soluble in sterile water, phosphate-buffered saline (PBS), or aqueous solutions; recommended reconstitution at 10-50 mg/mL for optimal stability |
| Storage | Store lyophilized powder at -20°C (-4°F); reconstituted solution stable at 2-8°C (36-46°F) for up to 7 days or -20°C for extended storage |
| pH Stability | Most stable at pH 3-4; susceptible to oxidation at neutral to alkaline pH |
| Redox State | Reduced form (GSH); contains free thiol group essential for antioxidant activity |
Note: Glutathione exists in two forms—reduced (GSH) and oxidized (GSSG). This product contains the reduced form, which provides active antioxidant function through its free sulfhydryl group. The tripeptide is sensitive to oxidation upon reconstitution, particularly in neutral or alkaline solutions, so researchers should prepare fresh working solutions or add stabilizers for extended use.
Proper handling and storage of glutathione is essential to maintain its reduced state, prevent oxidation, and ensure experimental reproducibility throughout research protocols.
Eternal Peptides provides comprehensive Certificates of Analysis (COAs) for every glutathione batch for transparency and traceability. A COA delivers detailed analytical data essential for experimental reproducibility, regulatory compliance, and quality assurance documentation.
That includes information on:
Eternal Peptides partners with leading independent analytical laboratories, including Janoshik and Finnrick Analytics, to conduct rigorous testing free from conflicts of interest. This third-party approach ensures objective, reliable results meeting international analytical standards.
For accessibility, COAs are lot-specific, uniquely identified by batch numbers printed on product vials, and readily accessible through the Lab Tests page on our website.
Synthetic glutathione is strictly for laboratory research purposes only and is not approved by the FDA or any regulatory authority for human consumption, veterinary use, clinical administration, therapeutic applications, or diagnostic procedures.
While glutathione is naturally present in the human body, the safety and efficacy of exogenous glutathione supplementation have not been fully established through controlled clinical trials meeting regulatory standards for therapeutic use. This product is intended exclusively for qualified researchers operating within approved protocols and appropriate biosafety frameworks.
Purchasers are solely responsible for ensuring compliance with all applicable federal, state, and local regulations governing research compounds, as well as institutional policies. Eternal Peptides does not condone, support, or provide guidance for any non-research applications of this compound.
https://pmc.ncbi.nlm.nih.gov/articles/PMC3303626/
https://pmc.ncbi.nlm.nih.gov/articles/PMC6523540/
https://pmc.ncbi.nlm.nih.gov/articles/PMC12046376/
https://pmc.ncbi.nlm.nih.gov/articles/PMC8716200/
https://pmc.ncbi.nlm.nih.gov/articles/PMC10017475/
https://pmc.ncbi.nlm.nih.gov/articles/PMC7173497/
https://pubs.acs.org/doi/10.1021/bi900030z
https://www.sciencedirect.com/science/article/pii/S2352304224002794
https://pmc.ncbi.nlm.nih.gov/articles/PMC3317707/
https://www.frontiersin.org/journals/immunology/articles/10.3389/fimmu.2019.00155/full
https://journals.plos.org/plosone/article?id=10.1371/journal.pone.0028378
https://pubmed.ncbi.nlm.nih.gov/12060676/
https://pmc.ncbi.nlm.nih.gov/articles/PMC2821140/
https://onlinelibrary.wiley.com/doi/full/10.1002/mef2.70026
https://pmc.ncbi.nlm.nih.gov/articles/PMC10762665/
https://pmc.ncbi.nlm.nih.gov/articles/PMC4684116/
https://bpspubs.onlinelibrary.wiley.com/doi/10.1111/bph.12847
https://pmc.ncbi.nlm.nih.gov/articles/PMC6188503/
https://onlinelibrary.wiley.com/doi/10.1111/jnc.70343
https://pmc.ncbi.nlm.nih.gov/articles/PMC3736736/
https://www.frontiersin.org/journals/pharmacology/articles/10.3389/fphar.2025.1737199/full
About the Author:
Dr. Sony Sherpa, MBBS, MD
is a board-certified clinician with a background in emergency medicine and clinical practice. She specializes in medical research analysis, ensuring that product information is grounded in evidence-based medicine and strictly adheres to the latest scientific standards in peptide research and recovery.