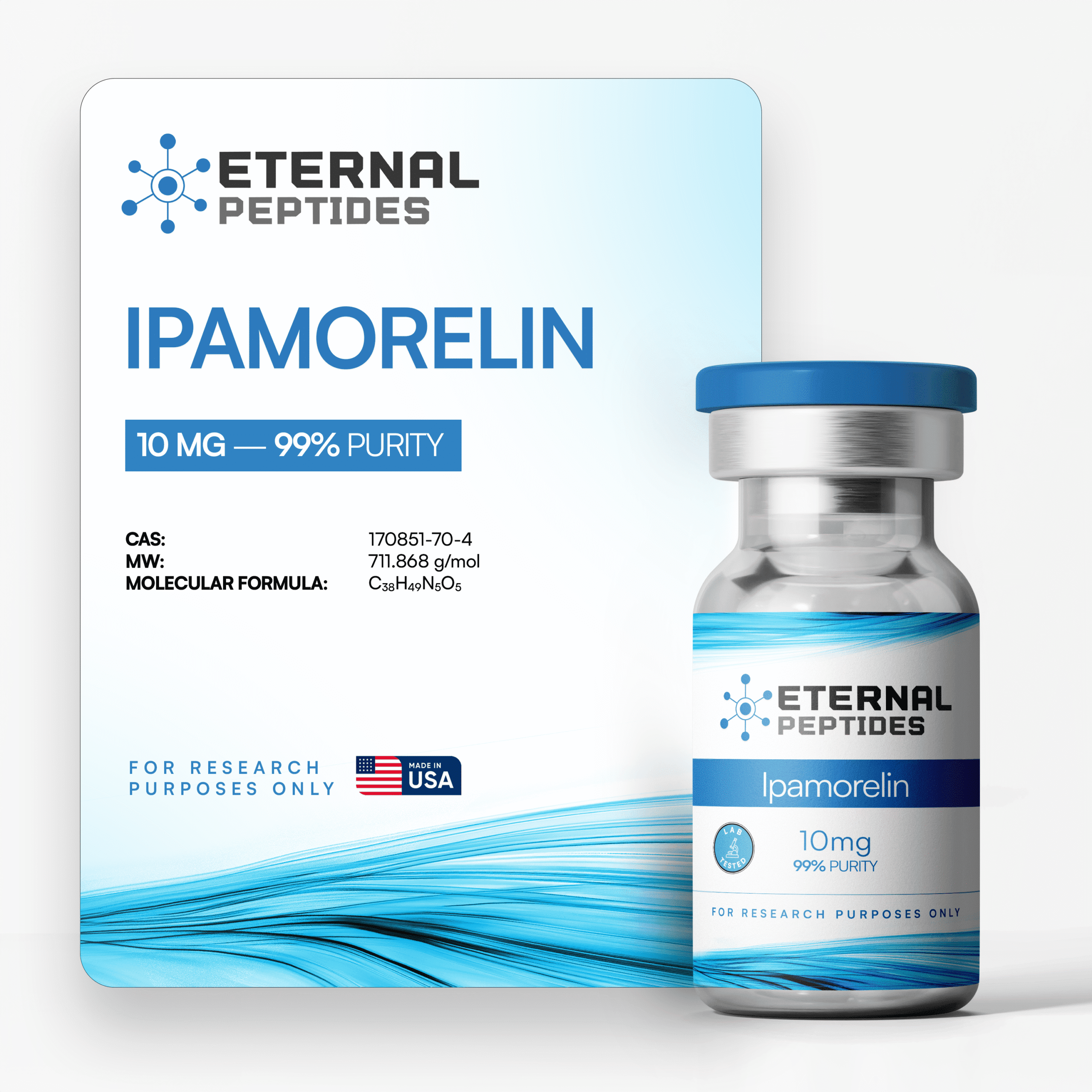
All products are handled with strict quality standards to ensure consistent research-grade excellence.
Our checkout is SSL encrypted and completely secure.
Our products are verified by independent third party laboratories to meet quality standards.
All product batches and lots are assigned unique identifiers and tied to publicly posted lab reports.
We are committed to helping our customers. If you have any questions or concerns, please reach out through our Contact Us page.
$189.99 Original price was: $189.99.$174.99Current price is: $174.99.
| Quantity | Discount | Price |
|---|---|---|
| 5 - 10 | 5% | $166.24 |
| 11 - 20 | 10% | $157.49 |
| 21+ | 15% | $148.74 |
Every batch of our research chemicals and peptides undergoes third-party testing.
*Disclaimer: This product is intended solely for laboratory research purposes. It is not suitable for consumption by humans, nor for medical, veterinary, or household purposes. Kindly review our Terms & Conditions before making a purchase.

Always quality-tested, verified with third party COA’s
At every step, we prioritize quality by conducting rigorous third-party testing on all our products. These tests focus on five key characteristics- identity, purity, sterility, and endotoxin levels, and heavy metal content-ensuring that each product meets the highest standards of quality with independent third-party Certificates of Analysis (COAS) to verify our commitment to excellence.
*Disclaimer: This product is intended solely for laboratory research purposes. It is not suitable for consumption by humans, nor for medical, veterinary, or household purposes.Kindly review our Terms & Conditions before making a purchase.
Shop the KLOW Blend (GHK-Cu, TB-500, BPC-157, KPV) in 80mg vials from Eternal Peptides, a leading peptides supplier known for unmatched quality. This formulation combines three peptides – GHK-Cu for tissue remodeling, TB-500 for cellular migration, BPC-157 for systemic stability, and KPV is for powerful anti-inflammation properties. Each vial is independently verified for purity and potency by leading labs including Janoshik and Finnrick Analytics, with COAs for every batch. Order KLOW Blend at competitive prices and get free USPS Priority shipping on orders over $200 with dedicated 24/7 U.S.-based support. For research use only.
KLOW Blend is a synergistic peptide formulation combining three well-characterized research compounds:
This strategic combination emerged from research exploring complementary mechanisms across tissue repair pathways. GHK-Cu is widely investigated for its roles in collagen synthesis and remodeling, TB-500 for cellular migration and angiogenesis, KPV for anti-inflammatory and antimicrobial potential, and BPC-157 for systemic stability and healing processes.
The scientific literature primarily investigates this blend for regenerative research applications, with extensive in vitro and animal model studies demonstrating potential synergistic effects across multiple tissue types.
Each component exhibits high aqueous solubility and stability in lyophilized form, making this formulation particularly suitable for controlled laboratory investigations. The combination allows researchers to study multi-pathway interactions in tissue repair mechanisms, cellular signaling cascades, and recovery processes within a single experimental framework.
KLOW Blend works through three complementary mechanistic pathways for its three peptide components that converge on tissue repair and cellular regeneration processes.
In rodent models and cellular assays, this combination has demonstrated accelerated wound closure, enhanced neovascularization in ischemic tissue models, and improved structural recovery following induced injury[1][2]. These mechanistic synergies suggest the blend addresses multiple phases of the healing cascade simultaneously, from initial inflammatory modulation through proliferative tissue formation and subsequent remodeling.
While each component has been studied extensively in preclinical contexts, controlled human clinical evidence for this specific combination remains limited. The observed effects in laboratory settings provide a foundation for ongoing investigational research, and therefore all these findings should be interpreted strictly within a preclinical experimental framework.
GHK-Cu functions as a copper delivery vehicle and signaling molecule, facilitating copper-dependent enzymatic reactions essential for extracellular matrix homeostasis. The tripeptide binds Cu²⁺ with high affinity, forming a stable complex that modulates lysyl oxidase activity, which is a copper-dependent enzyme critical for collagen and elastin cross-linking.
Research demonstrates that GHK-Cu influences gene expression across multiple pathways, with microarray studies revealing upregulation of genes associated with tissue repair and downregulation of inflammatory mediators[2]. The copper complex also stimulates fibroblast proliferation and migration in vitro, while promoting angiogenic factor expression in endothelial cell cultures.
In aged dermal fibroblast models, GHK-Cu treatment has restored more youthful gene expression patterns and increased synthesis of structural proteins. Animal studies utilizing excisional wound models show accelerated wound contraction and enhanced collagen deposition in GHK-Cu-treated groups compared to controls, suggesting its utility in tissue regeneration research[3].
TB-500, as a thymosin beta-4 analog, exerts its primary effects through sequestration of G-actin monomers, preventing their polymerization into F-actin filaments[4]. This actin-binding activity regulates cytoskeletal organization, facilitating cellular migration, differentiation, and survival under stress conditions.
In endothelial cell assays, TB-500 promotes tube formation and directional migration, which are hallmarks of angiogenic activity, by modulating focal adhesion dynamics and integrin signaling[6].
The peptide also upregulates vascular endothelial growth factor (VEGF) expression and enhances endothelial progenitor cell mobilization in animal models. In rodent myocardial infarction studies, TB-500 administration has been associated with increased capillary density in peri-infarct regions and improved ventricular function metrics[6].
Additional research in corneal injury models demonstrates accelerated epithelial migration and reduced inflammatory infiltration[7]. These cellular and tissue-level effects position TB-500 as a valuable tool for investigating migration-dependent repair processes and vascular remodeling mechanisms in controlled experimental settings.
BPC-157 demonstrates broad cytoprotective properties through modulation of growth factor signaling, particularly involving VEGF, EGF, and FGF pathways, as well as nitric oxide (NO) system interactions.
The peptide appears to stabilize vascular endothelium and promote angiogenesis through NO synthase pathway engagement, with studies showing both NO-dependent and NO-independent mechanisms depending on tissue context[1].
In gastrointestinal injury models, BPC-157 accelerates mucosal healing and reduces ulcer formation through enhanced blood flow and growth factor expression at injury sites.
Tendon and ligament damage models also reveal improved structural organization and biomechanical strength following BPC-157 administration, suggesting effects on collagen alignment and cross-linking.
BPC-157 exhibits neuroprotective properties in brain injury models, potentially through maintenance of blood-brain barrier integrity and modulation of neurotransmitter systems. Notably, BPC-157 demonstrates systemic effects when administered peripherally, with observed benefits in tissues distant from injection sites, indicating potential endocrine-like or circulation-mediated mechanisms that warrant further investigation in research applications.
KPV, a tripeptide fragment derived from the C-terminal region of α-melanocyte-stimulating hormone (α-MSH), exerts its primary effects through melanocortin receptor–mediated signaling, particularly MC1R and MC3R pathways expressed on immune and epithelial cells. Activation of these receptors leads to downstream suppression of pro-inflammatory transcription factors, most notably NF-κB, resulting in reduced expression of cytokines such as TNF-α, IL-6, and IL-1β.
At the cellular level, KPV attenuates inflammatory responses in macrophages and intestinal epithelial models while preserving tight junction integrity, supporting epithelial barrier function under inflammatory stress. In vitro studies demonstrate decreased chemokine production and reduced immune cell recruitment following KPV exposure, indicating a direct role in limiting localized inflammatory amplification[10].
Preclinical models of colitis and mucosal injury show that KPV administration is associated with reduced tissue edema, lower inflammatory infiltrate, and accelerated epithelial recovery[11].
Additional data suggest antimicrobial activity against select pathogenic organisms, potentially linked to membrane interactions and immune modulation[12]. Collectively, these mechanisms position KPV as a useful research peptide for studying inflammation-driven barrier dysfunction and melanocortin-regulated immune homeostasis in controlled experimental settings.
KLOW Blend’s synergistic formulation has attracted research interest across multiple domains, including tissue repair, musculoskeletal recovery, vascular health, and age-related degenerative processes.
The following applications represent observations from preclinical studies, primarily in rodent models and cellular assays. These findings do not imply human or veterinary therapeutic benefits, as controlled clinical evidence remains absent.
KLOW Blend is not approved for medical use, and Eternal Peptides does not promote or advocate for any human consumption or clinical applications.
Animal studies investigating wound healing have demonstrated that the combined mechanisms of KLOW Blend components accelerate closure rates and improve tissue quality in excisional wound models[1][2].
GHK-Cu’s matrix remodeling effects, TB-500’s promotion of cellular migration, and BPC-157’s vascular stabilization appear to address multiple phases of the healing cascade simultaneously. Rodent models also show enhanced collagen deposition, increased tensile strength in healed tissue, and reduced scar formation compared to untreated controls[2].
In these models, in vitro fibroblast cultures treated with this combination exhibit increased proliferation rates and organized extracellular matrix assembly.
Research in tendon, ligament, and muscle injury models suggests KLOW Blend components facilitate structural recovery and functional restoration. TB-500’s influence on cellular migration supports satellite cell recruitment to damaged muscle tissue, while BPC-157 has demonstrated improved biomechanical properties in injured tendons through enhanced collagen organization.
GHK-Cu contributes through copper-dependent cross-linking mechanisms that strengthen repaired connective tissue. Studies utilizing Achilles tendon transection models show improved load-bearing capacity and histological organization in treatment groups. Muscle contusion models reveal reduced inflammatory infiltration and accelerated functional recovery metrics[13].
The angiogenic properties observed across KLOW Blend components make this formulation valuable for vascular research applications. TB-500 promotes endothelial cell tube formation and VEGF expression, while BPC-157 stabilizes existing vasculature through nitric oxide pathway modulation. GHK-Cu enhances angiogenic factor expression in endothelial cultures.
Ischemic tissue models demonstrate increased capillary density and improved perfusion in treated animals. Myocardial infarction studies also show enhanced neovascularization in peri-infarct zones, though these effects remain strictly observational within experimental contexts[6][13].
Preclinical aging research has examined KLOW Blend components for their effects on tissue homeostasis and regenerative capacity in aged animal models. GHK-Cu demonstrates restoration of more youthful gene expression patterns in senescent fibroblasts, while TB-500 maintains cellular migration capacity that typically declines with age[2][14].
These laboratory studies in aged rodents show improved tissue repair kinetics and maintained vascular responsiveness compared to age-matched controls receiving standard care.
| Property | KLOW Blend Specification |
| Name | KLOW Blend: GHK-Cu (Glycyl-L-Histidyl-L-Lysine Copper Complex)
TB-500: (Thymosin Beta-4 Fragment) BPC-157: (Body Protection Compound-157) |
| Sequence | GHK-Cu: Gly-His-Lys-Cu²⁺
TB-500: 43-amino acid peptide analog BPC-157: Gly-Glu-Pro-Pro-Pro-Gly-Lys-Pro-Ala-Asp-Asp-Ala-Gly-Leu-Val |
| Molecular Weight | GHK-Cu: 401.91 g/mol (~340 Da)
TB-500: 889.01 g/mol (~4,963 Da) BPC-157: 1,419.54 g/mol (~1,419 Da) KPV: 342.43 g/mol |
| Molecular Formula | GHK-Cu: C14H22CuN6O4
TB-500: C38H68N10O14 BPC-157: C62H98N16O22 |
| PubChem CID | GHK-Cu: 378611
TB-500: 62707662 BPC-157: 9941957 KPV: 125672 |
| Product Form | Blue-purple lyophilized powder in sterile 80mg vials |
| Purity | ≥99% per component, verified by third-party testing (Janoshik, Finnrick Analytics) |
| Solubility | Highly soluble in sterile water, bacteriostatic water, or saline; reconstitute gently to preserve peptide integrity |
| Storage | Lyophilized: 35.6°-46.4°F (2-8°C); Reconstituted: 35.6-46.4°F (2-8°C) for up to 30 days, 68°F (-20°C) for extended storage |
Note: Each vial contains a total of 80mg of the combined peptide blend. Lot-specific Certificates of Analysis (COA) are available upon request for verification of purity, potency, and composition. This formulation is intended strictly for laboratory research purposes.
Store unopened KLOW Blend vials at 36-46°F (2-8°C) in a refrigerator, protected from light and moisture. For extended storage beyond 6 months, maintain at -4°F (-20°C) in a freezer. Ensure vials remain sealed until use to prevent degradation.
For the best stability, purity, and consistent results, get bacteriostatic water with your KLOW Blend order.
Once reconstituted, store solutions at 36-46°F (2-8°C) for up to 30 days. For longer-term storage, aliquot into single-use portions and store at -4°F (-20°C) or -112°F (-80°C). Divide reconstituted solutions into experiment-sized aliquots immediately after preparation to minimize freeze-thaw exposure.
For aliquoting, use sterile, low-binding microcentrifuge tubes and label with peptide identity, concentration, and preparation date.Avoid repeated freeze-thaw cycles, as these compromise peptide integrity and experimental reproducibility.
Handle all peptides using appropriate personal protective equipment (gloves, lab coat, safety glasses) in accordance with institutional biosafety protocols. Follow your organization’s chemical hygiene plan and dispose of materials per regulated waste guidelines.
Eternal Peptides provides lot-specific Certificates of Analysis (COAs) for every KLOW Blend product, ensuring traceability, reproducibility, and audit compliance for research applications. Each COA includes comprehensive analytical data obtained through third-party testing with leading independent laboratories, including Janoshik and Finnrick Analytics.
All test results are accessible on our lab tests page.
CoAs typically document:
KLOW Blend is supplied strictly for laboratory research purposes only. This product is not approved by the FDA or any regulatory authority for human use, veterinary use, clinical administration, therapeutic applications, or diagnostic procedures. Safety and efficacy in humans have not been established.
As the purchaser, you are solely responsible for ensuring compliance with all applicable local, state, federal, and international laws governing the acquisition, possession, and use of research peptides. All use must conform to institutional biosafety policies, research ethics guidelines, and appropriate laboratory protocols.
By purchasing KLOW Blend, you acknowledge that this product will be used exclusively for legitimate scientific investigation.
https://pmc.ncbi.nlm.nih.gov/articles/PMC12446177/
https://pmc.ncbi.nlm.nih.gov/articles/PMC6073405/
https://pmc.ncbi.nlm.nih.gov/articles/PMC4508379/
https://pmc.ncbi.nlm.nih.gov/articles/PMC4217450/
https://pubmed.ncbi.nlm.nih.gov/15037013/
https://pmc.ncbi.nlm.nih.gov/articles/PMC2701135/
https://pmc.ncbi.nlm.nih.gov/articles/PMC8275860/
https://pmc.ncbi.nlm.nih.gov/articles/PMC8504390/
https://www.mdpi.com/1424-8247/14/1/45
https://pmc.ncbi.nlm.nih.gov/articles/PMC5498804/
https://pmc.ncbi.nlm.nih.gov/articles/PMC2095288/
https://pmc.ncbi.nlm.nih.gov/articles/PMC6271067/
https://www.mdpi.com/2073-4409/10/6/1343
About the Author:
Dr. Sony Sherpa, MBBS, MD
is a board-certified clinician with a background in emergency medicine and clinical practice. She specializes in medical research analysis, ensuring that product information is grounded in evidence-based medicine and strictly adheres to the latest scientific standards in peptide research and recovery.