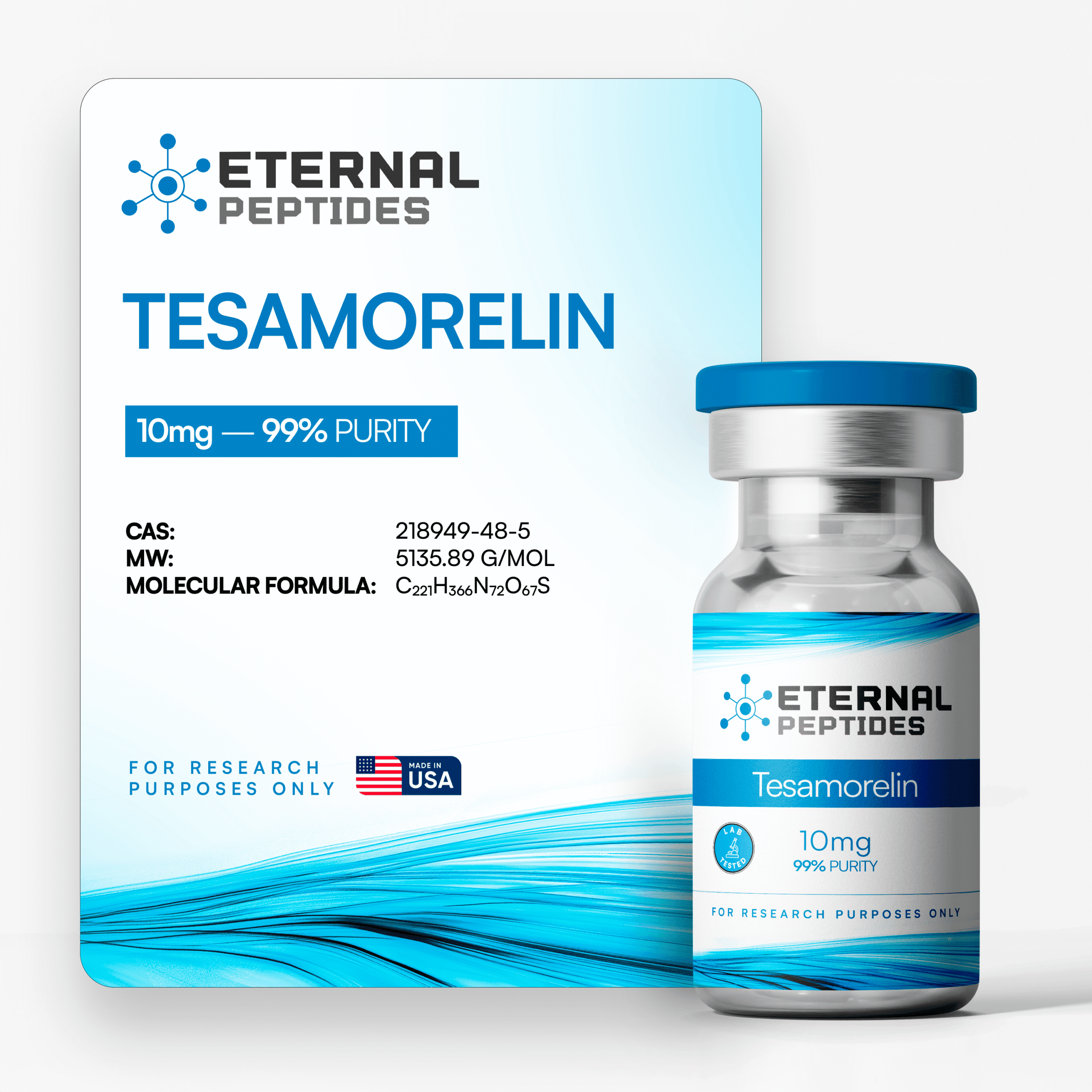
All products are handled with strict quality standards to ensure consistent research-grade excellence.
Our checkout is SSL encrypted and completely secure.
Our products are verified by independent third party laboratories to meet quality standards.
All product batches and lots are assigned unique identifiers and tied to publicly posted lab reports.
We are committed to helping our customers. If you have any questions or concerns, please reach out through our Contact Us page.
$34.99 – $59.99Price range: $34.99 through $59.99
| Quantity | Discount | Price |
|---|---|---|
| 5 - 10 | 5% | $56.99 |
| 11 - 20 | 10% | $53.99 |
| 21+ | 15% | $50.99 |
Every batch of our research chemicals and peptides undergoes third-party testing.
*Disclaimer: This product is intended solely for laboratory research purposes. It is not suitable for consumption by humans, nor for medical, veterinary, or household purposes. Kindly review our Terms & Conditions before making a purchase.

Always quality-tested, verified with third party COA’s
At every step, we prioritize quality by conducting rigorous third-party testing on all our products. These tests focus on five key characteristics- identity, purity, sterility, and endotoxin levels, and heavy metal content-ensuring that each product meets the highest standards of quality with independent third-party Certificates of Analysis (COAS) to verify our commitment to excellence.
*Disclaimer: This product is intended solely for laboratory research purposes. It is not suitable for consumption by humans, nor for medical, veterinary, or household purposes.Kindly review our Terms & Conditions before making a purchase.
Order KPV 10mg from Eternal Peptides, a trusted peptides provider with rigorous third-party testing by leading labs to ensure exceptional purity and reliable research outcomes. KPV is a tripeptide fragment derived from alpha-melanocyte stimulating hormone (α-MSH), designed for research in inflammatory response, gut barrier function, and immunomodulation studies. Get KPV 10mg vials at competitive pricing, with free USPS shipping for orders over $200 and dedicated support. KPV is sold for research use only.
KPV is a naturally occurring tripeptide consisting of the amino acid sequence Lysine-Proline-Valine (Lys-Pro-Val). It represents the C-terminal fragment of alpha-melanocyte stimulating hormone (α-MSH), a tridecapeptide hormone involved in various physiological processes including pigmentation, inflammation, and immune regulation.
KPV was identified as a biologically active fragment of α-MSH during research investigating the hormone’s anti-inflammatory properties in the 1980s and 1990s, when scientists discovered that this short sequence retained significant immunomodulatory activity without the pigmentation effects associated with the full-length hormone.
The scientific literature primarily investigates KPV for its potent anti-inflammatory and immunomodulatory properties, with research focusing on inflammatory bowel disease models, wound healing, antimicrobial activity, and epithelial barrier function. Studies examine its effects on cytokine production, NF-κB pathway inhibition, and mast cell stabilization across various inflammatory conditions.
Most published findings on KPV derive from in vitro cellular assays and animal models, particularly rodent studies of colitis and dermatitis, though some preliminary human data exists in dermatological applications.
For research use, KPV demonstrates excellent aqueous solubility and stability in physiological conditions, resisting rapid enzymatic degradation better than many longer peptide sequences.
KPV exerts its effects primarily through potent anti-inflammatory pathway modulation, particularly inhibition of nuclear factor kappa B (NF-κB) signaling, reduction of pro-inflammatory cytokine production, and stabilization of mast cell degranulation[1].
The tripeptide enters cells through endocytosis and peptide transport mechanisms, where it directly interferes with inflammatory transcription factor activation without requiring melanocortin receptor binding, distinguishing it from its parent hormone α-MSH.
Research demonstrates that KPV reduces production of tumor necrosis factor-alpha (TNF-α), interleukin-6 (IL-6), interleukin-8 (IL-8), and other inflammatory mediators in stimulated immune cells[2].
Also, animal studies of inflammatory bowel disease show reduced colonic inflammation, improved histological scores, and decreased disease activity indices following KPV administration[3]. Cellular assays reveal that KPV protects epithelial barrier integrity, reduces oxidative stress markers, and demonstrates antimicrobial properties against various pathogens, with wound healing models showing accelerated closure rates and improved tissue remodeling.
KPV’s primary anti-inflammatory mechanism involves direct inhibition of the nuclear factor kappa B (NF-κB) signaling pathway, a master regulator of inflammatory gene expression[1]. The tripeptide enters cells and interferes with the translocation of NF-κB from the cytoplasm to the nucleus, preventing the transcription of pro-inflammatory genes.
In vitro studies using lipopolysaccharide (LPS)-stimulated immune cells demonstrate that KPV treatment significantly reduces NF-κB activation, as measured by electrophoretic mobility shift assays and reporter gene assays[4].
This inhibition occurs in a dose-dependent manner and does not appear to involve melanocortin receptor activation, suggesting a novel mechanism distinct from α-MSH. Animal models of colitis show that KPV administration reduces nuclear NF-κB levels in intestinal tissue, correlating with decreased expression of downstream inflammatory mediators including COX-2, iNOS, and adhesion molecules[2].
The peptide’s ability to suppress NF-κB activation contributes to observed reductions in tissue inflammation, immune cell infiltration, and organ damage across multiple experimental inflammatory disease models.
KPV significantly reduces the production of pro-inflammatory cytokines from activated immune cells, including macrophages, dendritic cells, and T lymphocytes.
Cellular studies demonstrate that KPV treatment decreases TNF-α, IL-6, IL-8, IL-1β, and interferon-gamma (IFN-γ) secretion from cells stimulated with bacterial components, allergens, or other inflammatory triggers[1]. This cytokine reduction occurs at both transcriptional and post-transcriptional levels, with research showing decreased mRNA expression and reduced protein secretion.
Conversely, KPV appears to preserve or slightly enhance anti-inflammatory cytokine production, including IL-10, suggesting a rebalancing effect on immune responses rather than broad immunosuppression. In experimental colitis models, KPV-treated animals exhibit markedly reduced intestinal tissue levels of pro-inflammatory cytokines alongside improved histological scores and reduced clinical disease severity[2].
The cytokine-modulating effects extend to mast cells, where KPV prevents degranulation and histamine release in response to allergic triggers, as demonstrated in both isolated mast cell preparations and animal models of allergic inflammation.
Research demonstrates that KPV protects epithelial barrier integrity in intestinal, dermal, and respiratory tissues subjected to inflammatory challenges[2].
In cell culture models using intestinal epithelial monolayers, KPV treatment preserves tight junction protein expression and distribution, maintaining transepithelial electrical resistance (TEER) when cells are exposed to inflammatory cytokines or bacterial products. The peptide upregulates expression of tight junction proteins including occludin, claudins, and zonula occludens-1 (ZO-1), while preventing their degradation and redistribution under stress conditions.
Animal studies of inflammatory bowel disease show that KPV administration reduces intestinal permeability, as measured by reduced translocation of orally administered tracers into systemic circulation. This barrier-protective effect correlates with decreased bacterial translocation, reduced systemic endotoxin levels, and improved survival in severe colitis models.
Dermal wound healing studies reveal that KPV accelerates re-epithelialization, promotes organized collagen deposition, and enhances barrier restoration compared to control treatments, suggesting broad epithelial protective mechanisms across tissue types[5].
What this means is that KPV helps maintain the protective barrier of gut and skin tissues, keeping cells tightly connected and preventing harmful substances from crossing through damaged barriers in laboratory studies.
KPV demonstrates direct antimicrobial properties against various bacterial and fungal pathogens in microbiological assays, contributing to its protective effects in infection and inflammation models.
In vitro antimicrobial testing reveals that KPV inhibits growth of Staphylococcus aureus, Candida albicans, and certain gram-negative bacteria at physiologically relevant concentrations. The peptide appears to disrupt microbial membrane integrity and interfere with biofilm formation, mechanisms distinct from conventional antibiotics[6].
Research using infected wound models shows that topical KPV application reduces bacterial burden, accelerates healing, and decreases local inflammation compared to untreated controls.
In inflammatory bowel disease models where intestinal barrier compromise allows bacterial translocation, KPV treatment reduces bacterial presence in mesenteric lymph nodes and systemic organs, suggesting both direct antimicrobial effects and indirect benefits through barrier protection[7].
The antimicrobial activity occurs without apparent development of resistance in repeated exposure studies, though long-term resistance potential requires further investigation. This dual anti-inflammatory and antimicrobial profile makes KPV particularly interesting for research into conditions where inflammation and microbial dysbiosis interact.
While KPV demonstrates consistent anti-inflammatory and barrier-protective effects across multiple in vitro and animal model systems, controlled human clinical evidence remains extremely limited.
The majority of mechanistic insights and efficacy data derive from cellular assays and rodent studies, primarily using colitis, dermatitis, and wound healing models. Some preliminary human data exists for topical dermatological applications, but rigorous randomized controlled trials evaluating systemic KPV administration in human inflammatory conditions are absent from peer-reviewed literature.
The bioavailability, pharmacokinetics, optimal dosing, and safety profile of KPV in humans have not been systematically established through clinical research meeting current regulatory standards.
As such, researchers should interpret all findings within appropriate preclinical contexts and recognize that observed anti-inflammatory effects in controlled laboratory models do not automatically translate to human therapeutic efficacy or establish clinical safety profiles for any human applications.
KPV has generated significant research interest across inflammatory disease modeling, particularly in studies examining inflammatory bowel disease, dermatological inflammation, wound healing, allergic responses, and mucosal barrier function.
Since these applications emerge from observations in cellular assays, tissue culture systems, and rodent models investigating anti-inflammatory mechanisms and epithelial protection, they do not imply any established human therapeutic benefits or veterinary applications.
KPV is not approved by regulatory agencies such as the FDA for medical use, and Eternal Peptides does not promote, advocate for, or support any human consumption or clinical applications of this compound.
Animal studies using chemically induced colitis models demonstrate that KPV administration significantly reduces disease severity, as measured by clinical scoring systems, weight loss prevention, colon length preservation, and histological damage assessment[8].
Research employing dextran sodium sulfate (DSS) and trinitrobenzene sulfonic acid (TNBS) colitis protocols shows that KPV-treated rodents exhibit decreased inflammatory cell infiltration, reduced crypt damage, and lower ulceration scores compared to control groups[9].
The peptide reduces colonic tissue levels of pro-inflammatory cytokines including TNF-α, IL-6, IL-1β, and chemokines that recruit immune cells to inflamed tissue. Molecular studies reveal decreased NF-κB activation in intestinal epithelial cells and reduced myeloperoxidase activity, a marker of neutrophil infiltration.
In short, lab animals with experimentally induced intestinal inflammation show less severe disease, reduced tissue damage, and better-preserved gut barrier function when treated with KPV compared to untreated animals.
Dermatitis models in rodents demonstrate that topical or systemic KPV application reduces skin inflammation, epidermal thickening, immune cell infiltration, and clinical severity scores across various protocols, including contact hypersensitivity and atopic dermatitis-like conditions[10].
Research using hapten-induced contact dermatitis shows that KPV treatment decreases ear or skin swelling, reduces erythema, and lowers histological inflammation scores[4]. The peptide reduces local production of inflammatory mediators and decreases mast cell degranulation in affected skin tissues.
Similarly, in vitro studies using keratinocyte cultures reveal that KPV protects against inflammatory cytokine-induced damage, preserves cellular viability, and reduces oxidative stress markers. Psoriasis-like inflammation models demonstrate that KPV reduces epidermal hyperplasia, decreases parakeratosis, and normalizes keratinocyte differentiation markers in treated animals compared to vehicle controls.
In summary, these studies show that KPV treatment reduces swelling, redness, and tissue damage while protecting skin cells from inflammatory stress in culture dish experiments.
Experimental wound models reveal that KPV application accelerates healing kinetics, improves wound closure rates, and enhances tissue remodeling quality across dermal, oral mucosal, and intestinal wound paradigms. Research using full-thickness excisional wounds in rodents shows that KPV-treated wounds exhibit faster re-epithelialization, increased granulation tissue formation, and more organized collagen deposition during the remodeling phase.
The peptide reduces inflammatory cell persistence in wound beds while promoting appropriate angiogenesis and fibroblast activity. Diabetic wound models, which exhibit impaired healing, demonstrate particular responsiveness to KPV with significant improvements in healing rates and reduced chronic inflammation[11]. Intestinal anastomosis models show that perioperative KPV administration improves surgical wound healing, reduces anastomotic leakage rates, and decreases perianastomotic inflammation in experimental settings.
This means that, in lab animals, skin or internal wounds heal faster and with better-quality tissue formation when treated with KPV, including in diabetes models where healing is normally impaired.
Mast cell research demonstrates that KPV prevents degranulation and histamine release when cells are exposed to allergic triggers, both in isolated mast cell preparations and in animal models of allergic inflammation.
Studies using IgE-mediated mast cell activation protocols show that KPV pretreatment significantly reduces mediator release including histamine, leukotrienes, and pro-inflammatory cytokines[1].
Animal models of allergic airway inflammation reveal that KPV administration reduces bronchoalveolar lavage fluid cellularity, decreases airway hyperresponsiveness, and lowers lung tissue eosinophil infiltration following allergen challenge. The peptide modulates Th2-type immune responses, reducing IL-4, IL-5, and IL-13 production while maintaining balanced immune function.
KPV treatment may also help reduce anaphylactic responses in food allergy models, decreasing systemic histamine levels and improving survival rates in severe allergic reaction protocols.
Simply, KPV prevents allergic immune cells from releasing inflammation-causing chemicals, thus reducing allergic reactions in animal models of asthma and severe allergies.
| Property | Details |
| Name | KPV, Msh (11-13), (α-MSH C-terminal tripeptide fragment) |
| Sequence | Lys-Pro-Val (Lysine-Proline-Valine) |
| Molecular Formula | C₁₆H₃₀N₄O₄ |
| Molecular Weight | 342.43 g/mol |
| PubChem CID | 125672 |
| Product Form | Lyophilized powder in 10mg vials |
| Purity | ≥99%, verified via third-party laboratory analysis with lot-specific Certificates of Analysis (COA) available |
| Solubility | Highly soluble in sterile water, bacteriostatic water, or phosphate-buffered saline (PBS); recommended reconstitution at 1-5 mg/mL for optimal stability |
| Storage | Store lyophilized powder at -20°C (-4°F); reconstituted solution stable at 2-8°C (36-46°F) for up to 14 days or -20°C for extended storage |
| pH Stability | Stable across physiological pH range (6.0-8.0) |
| Receptor Binding | Does not require melanocortin receptor activation for anti-inflammatory activity |
Note: KPV represents the C-terminal tripeptide sequence of alpha-melanocyte stimulating hormone (α-MSH) and retains potent anti-inflammatory activity without the melanocortin receptor-mediated effects associated with the full-length hormone.
Proper handling and storage of KPV is essential to maintain peptide integrity, biological activity, and experimental reproducibility throughout research protocols.
Note: For optimal experimental outcomes and confidence in research material quality, order bacteriostatic water (sterile water) from Eternal Peptides with your KPV.
Eternal Peptides provides comprehensive Certificates of Analysis (COAs) for every KPV lot for transparency and traceability. Each COA delivers detailed analytical data essential for experimental reproducibility, regulatory compliance, and quality assurance documentation.
COA Contents:
Eternal Peptides partners with leading independent analytical laboratories, including Janoshik and Finnrick Analytics, to conduct rigorous independent testing. This means reliable results that meet international analytical standards and support publication-quality research.
Additionally, all COAs are lot-specific, uniquely identified by batch numbers printed on product vials, and readily accessible through the Lab Tests page on our website.
KPV is strictly for laboratory research purposes only and is not approved by the FDA or any regulatory authority for human consumption, veterinary use, clinical administration, therapeutic applications, or diagnostic procedures.
The safety and efficacy of KPV in humans have not been established through controlled clinical trials meeting regulatory standards. This product is intended exclusively for qualified researchers operating within institutional review board (IRB) approved protocols and appropriate biosafety frameworks.
Purchasers are solely responsible for ensuring compliance with all applicable federal, state, and local regulations governing research peptides, as well as institutional policies. Eternal Peptides does not condone, support, or provide guidance for any non-research applications of this compound.
https://pmc.ncbi.nlm.nih.gov/articles/PMC3403564/
https://pmc.ncbi.nlm.nih.gov/articles/PMC2431115/
https://pmc.ncbi.nlm.nih.gov/articles/PMC10582360/
https://pmc.ncbi.nlm.nih.gov/articles/PMC2095288/
https://www.medsci.org/v22p4175.htm
https://pmc.ncbi.nlm.nih.gov/articles/PMC7533533/
https://pmc.ncbi.nlm.nih.gov/articles/PMC11357942/
https://pmc.ncbi.nlm.nih.gov/articles/PMC4957955/
https://pmc.ncbi.nlm.nih.gov/articles/PMC11063560/
https://www.sciencedirect.com/science/article/abs/pii/S0022354917301740
https://pmc.ncbi.nlm.nih.gov/articles/PMC3708299/
About the Author:
Dr. Sony Sherpa, MBBS, MD
is a board-certified clinician with a background in emergency medicine and clinical practice. She specializes in medical research analysis, ensuring that product information is grounded in evidence-based medicine and strictly adheres to the latest scientific standards in peptide research and recovery.